The Amazing Musculatory System
Muscles are the tissues that allow our bodies to move! The more you know about how your muscles work, the better you’ll be able to apply it to your life – such as lifting weights. To get you started, we created this short basics video.
There are three different types of muscle: cardiac, smooth, and skeletal. Each is responsible for a different function.
Cardiac Muscle
Cardiac muscle is striated and can produce impulses and contract spontaneously. Heart cells, or myocardial cells, are short, branched, and interconnected. Each myocardial cell is joined to adjacent myocardial cells by gap junctions, which are electrical synapses. Since all the cells in a myocardium (a mass of myocardial cells) are electrically connected, a myocardium acts as a single functional unit. This means that a myocardium contracts to its full potential each time because all of its cells contribute to the contraction.
Smooth Muscle
Smooth muscle is found in the walls of blood vessels and bronchioles, the digestive tract, the ureters, the ductus deferentia, and the uterine tubes. Smooth muscle is not striated (meaning it does not contain sarcomeres). This is because smooth muscle cells are required to contract even when greatly stretched, such as in the urinary bladder.
Now that we’re familiar with cardiac and smooth muscle, let’s focus primarily on skeletal muscle.
Skeletal Muscle
Skeletal muscle is a biological machine with the primary function of converting chemical energy (stored in the bonds of ATP) into mechanical work. Skeletal muscles are usually attached to bones via tendons. When a muscle contracts, tension is placed upon the tendons and the attached bones. This tension causes movement of the bones at a joint. Skeletal muscle is also a great source of heat, a dynamic metabolic store, and a source of protective padding.
Composition
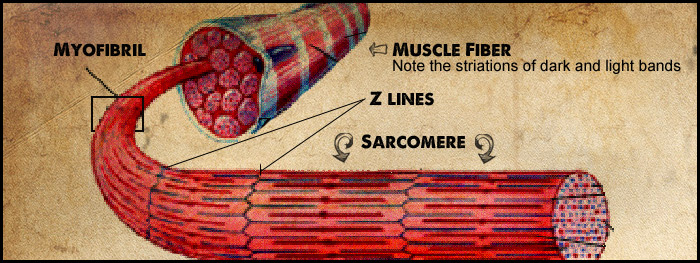
When viewed under a microscope, muscle fibers appear to be striated. These striations are produced by alternating dark and light bands that span the width of the muscle fiber. The dark bands are called A bands, and the light bands are called I bands. The A bands are composed of thick filaments, and the I bands are composed of thin filaments. When muscle fibers are viewed at a high magnification through an electron microscope, thin dark lines can be seen in the middle of the I bands. These are called Z lines (or Z discs). The arrangement of thick and thin filaments between a pair of Z lines forms a repeating pattern that is the basic subunit of striated muscle. These subunits are called sarcomeres.
Muscle Contraction
Muscle is 75% water and 20% protein. The protein in muscle is mostly myosin (thick filaments) and actin (thin filaments), which are contractile proteins that play a big role in muscle contraction. When a muscle contracts it decreases in length due to the shortening of individual fibers. This occurs as a result of the shortening of the distance from Z disc to Z disc. As the sarcomere shortens in length, the A bands do not shorten, although the I bands do. This shortening is actually produced by the sliding of the thin filaments over the thick filaments. This is known as the Sliding Filament Theory. In other words, the bands shorten, the filaments do not.
Cross Bridges
The action of cross bridges results in the sliding of filaments. Cross bridges are part of the myosin proteins that extend from the axis of the thick filaments to form ‘arms’ that terminate in globular ‘heads.’ Each globular head contains an ATP-binding site that is associated with an actin-binding site. The globular head hydrolyzes (splits) ATP into ADP and inorganic phosphate (Pi). When ATP is hydrolyzed, the mysoin head is in the ready state, and thus has the potential energy required for contraction. After the myosin head binds to actin, myosin undergoes a conformational change, causing the cross bridge to produce a power stroke. The power stroke is the force that pulls the thin filaments over the thick filaments. After the power stroke, the bound ATP is released and a new ATP molecule binds to the myosin head. If ADP was not released and a new ATP molecule did not bind to myosin, then the myosin heads would remain bound to actin, resulting in rigor mortis. In rigor mortis, the muscles stiffen until myosin and actin start to decompose, such as after death.
Confusing? Let’s break this down.
- A fiber is at rest and a cross bridge is not attached to actin; ATP has been hydrolyzed.
- A cross bridge binds to actin.
- Pi is released causing a conformational change in the myosin head.
- A power stroke causes the thin filaments to slide of the thick filaments; ADP is released.
- A new ATP binds to the myosin head which allows it to release from actin.
- ATP is hydrolyzed, causing the cross bridge to return to its original orientation…
- Back to 1.
Control of Muscle Contraction
When a muscle is at rest, the calcium concentration in the sarcoplasm (a muscle cell’s cytoplasm) is very low and cross bridges are prevented from attaching to actin. For a muscle to contract, high concentrations of calcium are needed. Action potentials are conducted into the muscle fiber which stimulate the opening of calcium-release channels in the sarcoplasmic reticulum (smooth ER found in muscle cells), causing calcium to diffuse into the sarcoplasm and stimulate contractions. When the action potential ends, the calcium-release channels in the sarcoplasmic reticulum close, allowing calcium to be actively transported out of the sarcoplasm and sarcomeres. This allows the muscle to relax. Without the removal of calcium, the muscle cannot relax.
Adaptations of Muscles to Exercise
Muscle cells grow, or hypertrophy, due to frequent bouts of high-intensity resistance training, such as weight lifting. Hypertrophy means muscle cells grow in size, not in number. Eating a sufficient amount of protein is also important for building muscle.
The opposite of hypertrophy is atrophy, or the reduction in muscle mass. This occurs if a muscle is not being used, such as a broken leg in a cast. Atrophy also occurs as a person ages. Even in health, aging is associated with a progressive decline in muscle function. However, the good news is that muscle remains responsive to resistance training even very late in life, meaning, it is possible to build muscle at an old age.
Related Topics
Muscles are the tissues that allow our bodies to move! The more you know about how your muscles work, the better you’ll be able to apply it to your life – such as lifting weights. To get you started, we created this short basics video.
There are three different types of muscle: cardiac, smooth, and skeletal. Each is responsible for a different function.
Cardiac Muscle
Cardiac muscle is striated and can produce impulses and contract spontaneously. Heart cells, or myocardial cells, are short, branched, and interconnected. Each myocardial cell is joined to adjacent myocardial cells by gap junctions, which are electrical synapses. Since all the cells in a myocardium (a mass of myocardial cells) are electrically connected, a myocardium acts as a single functional unit. This means that a myocardium contracts to its full potential each time because all of its cells contribute to the contraction.
Smooth Muscle
Smooth muscle is found in the walls of blood vessels and bronchioles, the digestive tract, the ureters, the ductus deferentia, and the uterine tubes. Smooth muscle is not striated (meaning it does not contain sarcomeres). This is because smooth muscle cells are required to contract even when greatly stretched, such as in the urinary bladder.
Now that we’re familiar with cardiac and smooth muscle, let’s focus primarily on skeletal muscle.
Skeletal Muscle
Skeletal muscle is a biological machine with the primary function of converting chemical energy (stored in the bonds of ATP) into mechanical work. Skeletal muscles are usually attached to bones via tendons. When a muscle contracts, tension is placed upon the tendons and the attached bones. This tension causes movement of the bones at a joint. Skeletal muscle is also a great source of heat, a dynamic metabolic store, and a source of protective padding.
Composition
When viewed under a microscope, muscle fibers appear to be striated. These striations are produced by alternating dark and light bands that span the width of the muscle fiber. The dark bands are called A bands, and the light bands are called I bands. The A bands are composed of thick filaments, and the I bands are composed of thin filaments. When muscle fibers are viewed at a high magnification through an electron microscope, thin dark lines can be seen in the middle of the I bands. These are called Z lines (or Z discs). The arrangement of thick and thin filaments between a pair of Z lines forms a repeating pattern that is the basic subunit of striated muscle. These subunits are called sarcomeres.
Muscle Contraction
Muscle is 75% water and 20% protein. The protein in muscle is mostly myosin (thick filaments) and actin (thin filaments), which are contractile proteins that play a big role in muscle contraction. When a muscle contracts it decreases in length due to the shortening of individual fibers. This occurs as a result of the shortening of the distance from Z disc to Z disc. As the sarcomere shortens in length, the A bands do not shorten, although the I bands do. This shortening is actually produced by the sliding of the thin filaments over the thick filaments. This is known as the Sliding Filament Theory. In other words, the bands shorten, the filaments do not.
Cross Bridges
The action of cross bridges results in the sliding of filaments. Cross bridges are part of the myosin proteins that extend from the axis of the thick filaments to form ‘arms’ that terminate in globular ‘heads.’ Each globular head contains an ATP-binding site that is associated with an actin-binding site. The globular head hydrolyzes (splits) ATP into ADP and inorganic phosphate (Pi). When ATP is hydrolyzed, the mysoin head is in the ready state, and thus has the potential energy required for contraction. After the myosin head binds to actin, myosin undergoes a conformational change, causing the cross bridge to produce a power stroke. The power stroke is the force that pulls the thin filaments over the thick filaments. After the power stroke, the bound ATP is released and a new ATP molecule binds to the myosin head. If ADP was not released and a new ATP molecule did not bind to myosin, then the myosin heads would remain bound to actin, resulting in rigor mortis. In rigor mortis, the muscles stiffen until myosin and actin start to decompose, such as after death.
Confusing? Let’s break this down.
- A fiber is at rest and a cross bridge is not attached to actin; ATP has been hydrolyzed.
- A cross bridge binds to actin.
- Pi is released causing a conformational change in the myosin head.
- A power stroke causes the thin filaments to slide of the thick filaments; ADP is released.
- A new ATP binds to the myosin head which allows it to release from actin.
- ATP is hydrolyzed, causing the cross bridge to return to its original orientation…
- Back to 1.
Control of Muscle Contraction
When a muscle is at rest, the calcium concentration in the sarcoplasm (a muscle cell’s cytoplasm) is very low and cross bridges are prevented from attaching to actin. For a muscle to contract, high concentrations of calcium are needed. Action potentials are conducted into the muscle fiber which stimulate the opening of calcium-release channels in the sarcoplasmic reticulum (smooth ER found in muscle cells), causing calcium to diffuse into the sarcoplasm and stimulate contractions. When the action potential ends, the calcium-release channels in the sarcoplasmic reticulum close, allowing calcium to be actively transported out of the sarcoplasm and sarcomeres. This allows the muscle to relax. Without the removal of calcium, the muscle cannot relax.
Adaptations of Muscles to Exercise
Muscle cells grow, or hypertrophy, due to frequent bouts of high-intensity resistance training, such as weight lifting. Hypertrophy means muscle cells grow in size, not in number. Eating a sufficient amount of protein is also important for building muscle.
The opposite of hypertrophy is atrophy, or the reduction in muscle mass. This occurs if a muscle is not being used, such as a broken leg in a cast. Atrophy also occurs as a person ages. Even in health, aging is associated with a progressive decline in muscle function. However, the good news is that muscle remains responsive to resistance training even very late in life, meaning, it is possible to build muscle at an old age.